Solubility
|
We started talking about solubility with mixtures. Mixtures can be separated by physical means.
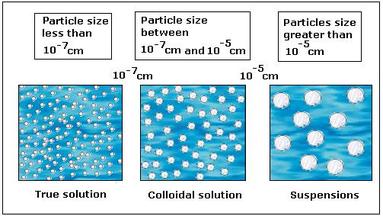
Solutions are homogeneous, and the tiny particles in the solvent are equally distributed. Suspensions are heterogeneous, and must be constantly stirred so the particles do not settle. Colloids look homogeneous like a solution, but their particles are slightly larger than those of a solution, sometimes giving a cloudy appearance. The Tyndall Effect is used to differentiate between a solution and a colloid. Click here for the notes on mixtures. |
|
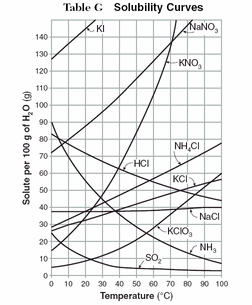
To the left is a solubility curve. This is a graph that compares how much of different solutes can be dissolved in 100 g of water at various temperatures. When reading the graph, anything above a solute's line is a supersaturated solution, anything at the line is a saturated solution, and anything below the line is an unsaturated solution.
Click here for help on reading solubility curves. |